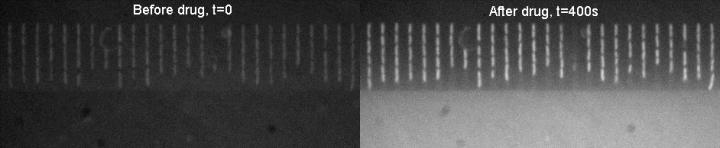
Drug uptake in bacteria before and after. Courtesy of Dr. Stefano Pagliara, University of Exeter
A new technique could help reduce antibiotic prescribing by predicting which drugs could be effective in fighting bacteria within minutes.
Scientists at the University of Exeter have developed the method, which allows users to see whether a bacterium is likely to respond to antibiotics. The research is currently in early stages of development, and the team hope the miniaturized devices they use for this research could one day be based in clinics, reducing the number of different antibiotics prescribed to patients.
The technique works by examining whether fluorescent qualities of the antibiotics are taken up by bacteria. If so, the bacteria glow brighter under the microscope, revealing that the antibiotic has infiltrated the membrane and could be effective. The research, published in the journal Lab on a Chip, could contribute to efforts to reduce prescribing, and also enable the development of more effective antibiotics, to help fight the global threat of antibiotic resistance.
Antibiotic resistance is recognized as a major global threat. As these drugs increasingly fail to work, around 10 million people are predicted to die annually of infections by 2050.
The new technique uses a special microscope and a miniaturized device into which a sample of the bacteria is injected, along with the antibiotic. To date, the team has used the antibiotic ofloxacin, which glows fluorescent under ultraviolet light. Bacteria also glow when the antibiotic is taken up. However, if they remain dark, the antibiotic has no chance of working and killing the bacteria.
Dr. Stefano Pagliara, a biophysicist in the Living Systems Institute, leading this research at the University of Exeter, said, "We're really excited about the potential for this technique to make a meaningful reduction in prescribing, helping to fight the global threat of antibiotic resistance. At the moment, it can take days for clinicians to get a lab result, which involves growing bacteria, but there is still some guess work involved. Our technique could reduce the use of multiple antibiotics to try and fight a bacterial infection."
Dr. Jehangir Cama, an industry research fellow at the Living Systems Institute, who performed the experimental work of this research, said, "Our next step is to further develop this exciting new method by combining it with more advanced microscopy techniques, to see where exactly the antibiotics go when they enter the bacteria."
The team is now working on expanding the technique, by manipulating the fluorescent qualities of other forms of antibiotics so they can work in the same way. Further research in this area has been funded by QUEX, a partnership between the University of Exeter and The University of Queensland in Australia. The Queensland team, led by Dr. Mark Blaskovich, director of the Centre for Superbug Solutions at the Institute for Molecular Bioscience, is developing fluorescent versions of other antibiotics so they can be tested in a similar manner.
Blaskovich adds, "I am enthused about the opportunities to improve our fundamental understanding of the interactions between antibiotics and bacteria and how this leads to antimicrobial resistance, by combining our novel antibiotic-derived probes with the cutting edge single cell analysis capabilities of the Exeter group."
This work will be carried out in collaboration with Prof Krasimira Tsaneva-Atanasova, a mathematician at the University of Exeter. The combined teams are currently seeking a jointly-supervised PhD student, funded by a Queensland-Exeter (QUEX) PhD scholarship.
The paper is titled 'Single-cell microfluidics facilitates the rapid quantification of antibiotic accumulation in Gramnegative bacteria.'
Source: University of Exeter
852032 574863I havent checked in here for some time as I thought it was obtaining boring, but the last couple of posts are wonderful quality so I guess Ill add you back to my everyday bloglist. You deserve it my friend 7015