A healthcare worker demonstrates the portable negative pressure procedural tent. Courtesy of the University of Michigan Center for Integrative Research in Critical Care.
Treating patients with COVID-19 means healthcare workers are putting their own health and safety on the line each day. Now, two new devices could offer additional safety for healthcare workers, while allowing more effective treatment for patients with infections spread by respiratory droplets.
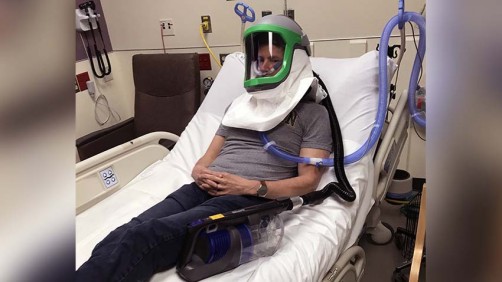
In two new proof-of-concept papers, fast-tracked and published in the International Journal of Tuberculosis and Lung Disease, researchers from the University of Michigan Center for Integrative Research in Critical Care (MCIRCC) describe two devices, dubbed Aerosolve, they designed: a portable negative pressure helmet and a portable negative pressure procedural tent.
“We saw a need for devices that could protect our fellow health care workers and enable alternative treatment methods for patients with COVID-19, instead of defaulting to a ventilator,” says Kevin Ward, MD, a professor of emergency medicine and executive director of MCIRCC.
“Both the helmet and tent create a portable negative pressure environment, potentially mitigating the need for creation of additional negative pressure rooms,” says Benjamin Bassin, MD, an assistant professor of emergency medicine at Michigan Medicine, director of the Joyce and Don Massey Family Foundation Emergency Critical Care Center (EC3), and one of the team members leading the projects. “This is important because we’ve seen shortages of ventilators and negative pressure rooms in areas across the world that were hit hard by the virus.”
Bassin explains that the helmet allows for healthcare workers to liberally use heated high flow nasal cannula (HFNC), a therapy of up to 60 liters of oxygen per minute to the nasal and oral cavity, non-invasive ventilation or nebulized medications. Using these treatments may spare or delay the need for a mechanical ventilator or potentially allow earlier transition of a patient from mechanical ventilation.
“The helmet design is compact and creates and maintains a personal negative pressure environment that the patient can wear during transport, imaging and testing,” Bassin says.
The portable negative pressure procedural tent circulates and removes air—including droplets exhaled by the patient—through an attached vacuum motor and high efficiency particulate air (HEPA) filter, clearing it of the virus before being released into the room.
“The tent is unique in that it allows us to access and perform a greater number of procedures the patient may need compared to the helmet,” says Nathan Haas, MD, an instructor of emergency medicine at Michigan Medicine and member of the project team. “The entire apparatus is disposable and can be used with a single patient, with the exception of the manifold base which can be cleaned and reused.”
The research team says one challenge with the heated high flow nasal cannula oxygen treatment, and some other respiratory therapies, is that they may result in aerosolization, or the production of airborne particles and tiny liquid droplets of the virus, putting healthcare workers at a higher risk of exposure.
“Clinicians were concerned about the use of heated high flow nasal cannula and non-invasive ventilation in patients with COVID-19 due to potential risk of aerosolization, so some avoided those therapies and put patients on ventilators early,” Bassin says. “We were potentially not using an additional tool in our arsenal against the disease that could help patients because of the risks to health care workers.”
Haas says the devices are envisioned to be used for other infectious diseases, such as tuberculosis and influenza.
“These devices allow more liberal use of proven therapies, while potentially lowering the risk of exposure to health care workers by containing and filtering respiratory droplets and aerosols,” he says.
The devices were created in a collaboration between the University of Michigan and FlexSys Inc. in Ann Arbor, Michigan.
FlexSys Inc. developed prototypes of the devices at its own cost and the first 50 tent units will be delivered to Michigan Medicine by the end of this week for use in the hospital.
"My only goal is to put as many of these devices in the hands of caregivers as quickly as possible," said Sridhar Kota, PhD, the Herrick Professor of Engineering at the U-M College of Engineering and founder of FlexSys Inc., who co-led the development of the devices.
U-M and FlexSys Inc. are seeking commercial partners to supply the tents and helmets to hospitals as soon as possible, and are extending non-exclusive royalty-free licenses to companies for the duration of the COVID-19 pandemic.
“We hope these devices allow our fellow healthcare workers to safely and effectively treat patients,” Ward says.
Disclosure: FlexSys Inc. is a startup from the University of Michigan, and Sridhar Kota holds equity in the company.
Source: Michigan Medicine - University of Michigan
Be the first to comment on "Two New Devices Protect Healthcare Workers, Help Patients During COVID-19"