Intestinal micro-biodiversity is important for maintaining colorectal and general health. Imbalance in this microenvironment can result in chronic infections, even cancer. Development of colorectal cancer (CRC) has been alluded to abnormal gut-bacterial activity and infections. However, the complexity of the intestinal microbiome makes it challenging to identify which bacterial types promote CRC pathogenesis in what way.
In a recent review, published in the Chinese Medical Journal on February 20, investigates this using chronic Salmonella infections as an example. Dr. Jun Sun, author, and professor at the University of Illinois at Chicago, explains the study’s rationale. “Better understanding of the enteric microbiome and pathogens could lead to new approaches for preventing and treating CRC, by inhibiting chronic infection and inflammation.”
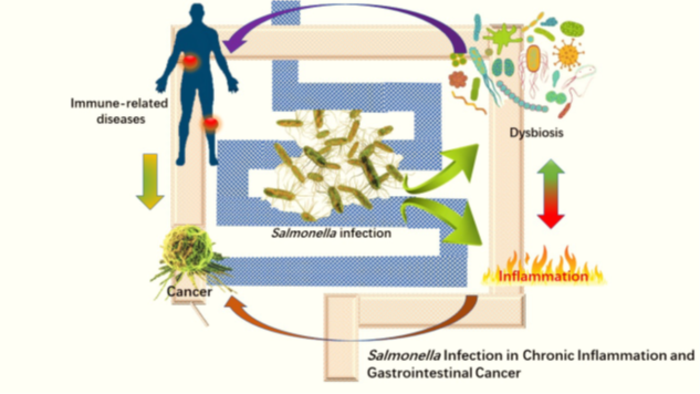
Pathogenic bacteria attack host physiology by interacting with host cells and other gut microbes, either physically or chemically (by releasing bacterial proteins and metabolites). These interactions can lead to disrupted immune responses; chronic intestinal infections; inflammation; DNA damage; and eventually, carcinogenesis.
For instance, Salmonella infections can have varied effects, from mild gastroenteritis to chronic irritable bowel disease (IBD)—a known risk factor for CRC. Patients with IBD or prior Salmonella infection are more likely to develop CRC than the general populace. This likelihood is further increased in patients with preexisting genetic susceptibilities, cell signaling defects, or mutations.
Bacterial infections can also impair the host’s immune response by controlling inflammation and modifying key signaling pathways. Bacterial effectors, like AvrA found in Salmonella, either promote or suppress inflammation in order to create a hospitable environment for the bacteria’s survival, and the maintain chronic infection. Studies suggest an increased occurrence of CRC tumorigenesis in mice infected with AvrA+ Salmonella compared to uninfected mice. Clinical data also indicate the presence of bacterial AvrA in the colorectal mucosa of CRC patients as against non-CRC patients. Similarly, Salmonella interacts with host epithelial and immunological cells to regulate cell signaling and modify chromosomal material, compromising the host’s immune response while promoting CRC progression. Moreover, these bacterial activities significantly alter the intestinal microenvironment, making it conducive for tumorigenesis.
While lab studies have progressed our understanding of these mechanisms, there is still need for human data to confirm the exact relation between bacterial infections and CRC development. “Looking forward, we hope for new insights in this field that can help illustrate how enteric bacteria control tumorigenesis and identify novel techniques to target specific pathways that influence CRC," says Sun.